The two companies expect to work together to quickly respond to the shortage of critical ventilation equipment necessary to support patients breathing. Assuming a stable supply of essential parts, the companies will be rapidly scaling up production from approximately 40,000 ventilators in April to between 150,000 and 200,000 ventilators a month by June. Together, Xerox and Vortran could produce as many as 1 million ventilators in the coming months.
While the GO2Vent is not a replacement for ventilators found in intensive care units (ICUs), it is widely used in emergency situations, inter-hospital transport and MRIs. Given the shortage of ICU-grade ventilators, medical professionals are utilizing tools like this and other technology to support patients who do not yet or no longer need an ICU-level breathing device, which can be freed up for another patient.
Our smartest minds met (virtually) with Vortran’s smartest minds and figured out how to mass produce this critical technology. We want to help make sure doctors, nurses and paramedics on the frontlines have the resources they need to help the rising number of patients with COVID-19.
John Visentin, vice chairman and chief executive officer, Xerox
Xerox plans to manufacture these FDA-approved ventilators and APM-Plus devices at its facility outside of Rochester, NY, where the company was founded and maintains a large presence. Vortran will continue to manufacture ventilators at its current facility in Sacramento, CA.
The partnership with Xerox has one clear goal – to help save as many lives as possible. With Vortran’s proven technology and Xerox’s ability to hyper-scale manufacturing, we believe we can supply healthcare providers as many as 1 million ventilators in the coming months. For all of us, this will be the most important thing we ever do.
Gordon A. Wong, M.D, Vortran Co-founder and CEO
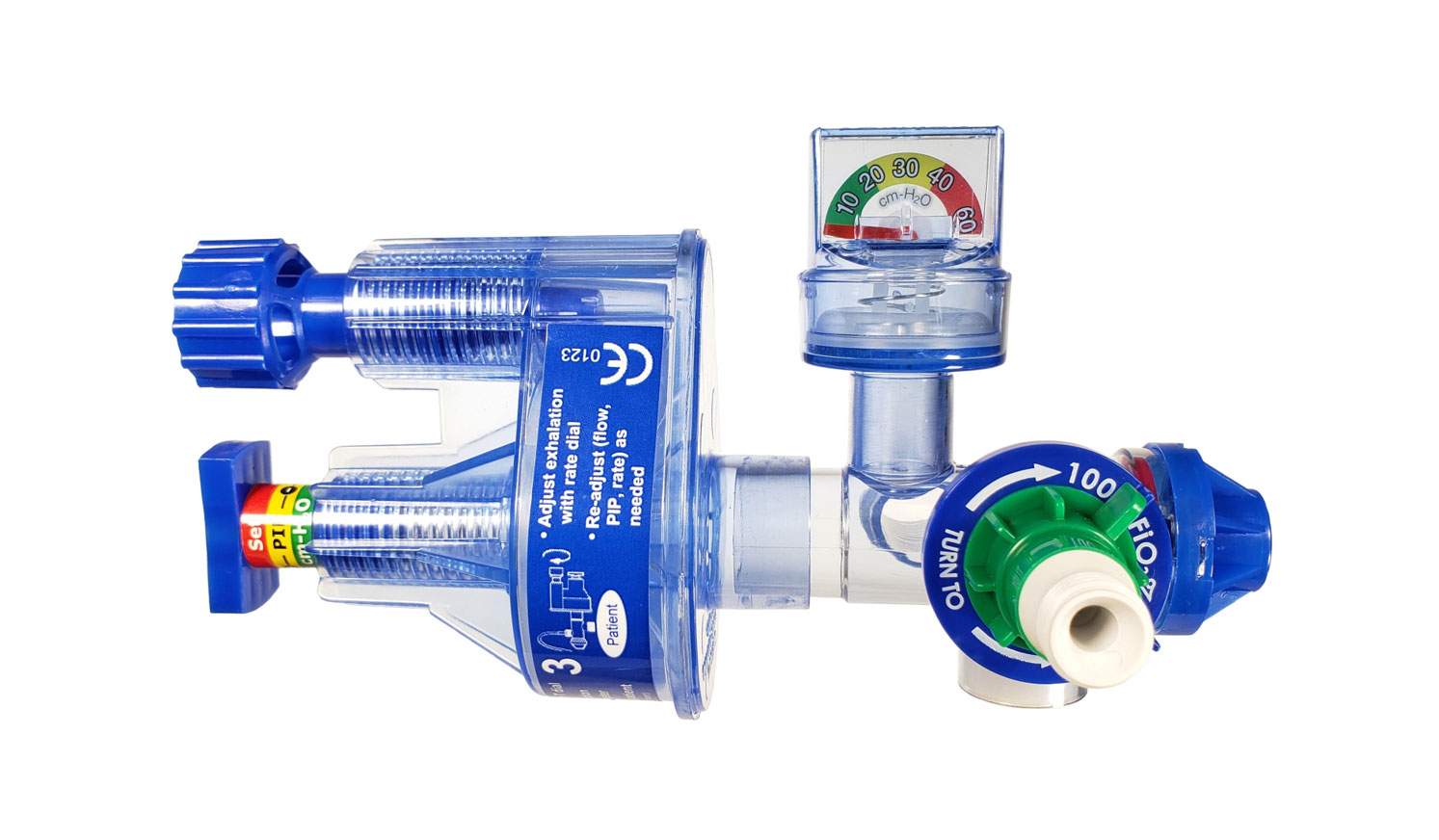
Vortran’s GO2Vent was designed for emergency use, natural disasters and disease outbreaks such as the COVID-19 pandemic. A gas-operated, disposable ventilator that can be set-up within minutes and discarded after use by a single patient, it provides support via a secure airway and can be operated on a compressor, oxygen or air with a minimum of 10 liters per minute flow rates.
The GO2Vent can provide continuous ventilatory support for up to 30 days and operates independent of Vortran’s APM-Plus. The APM-Plus is a battery operated, portable device that connects to a GO2Vent and provides enhanced monitoring of patient status and key respiratory parameters. Given the need to confirm key supplies, the companies are actively assessing how quickly production can ramp for the APM-Plus devices.
In addition to scaling up production of the GO2Vent and APM-Plus, Xerox and Vortran expect to compile and analyze data and feedback from healthcare professionals on the frontlines of the battle against COVID-19 in order to design and mass produce external, in-line modifications that can be added to the GO2Vent to expand the potential applications of this life-saving equipment.